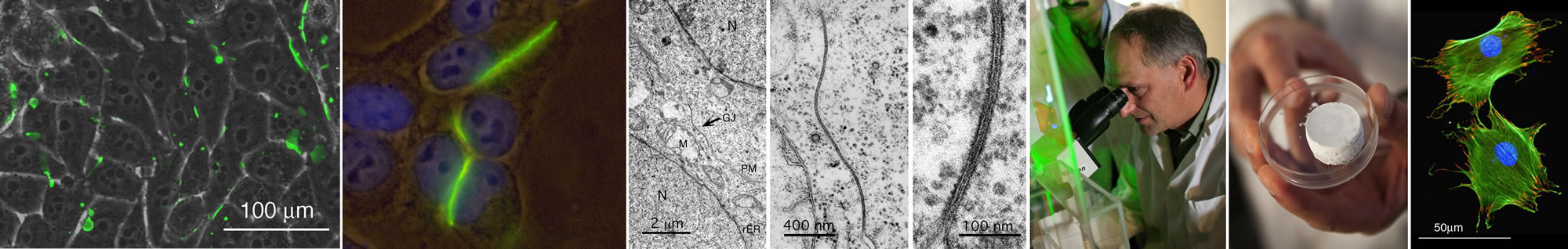
We investigate the structure and function of gap junction membrane channels, and the biological characteristics of novel bio-scaffolds made of dual-porous bioactive glasses.
Cells of multi-cellular organisms are autonomous units, yet constantly depend on signals from their surroundings. Signals can either be transmitted between cells and the extra-cellular milieu, or directly from cell to cell. Information exchange is mediated by membrane proteins that assemble into localized, spatial, and temporal organized multiunit transmembrane protein complexes, such as tight junctions, adherens junctions, desmosomes, 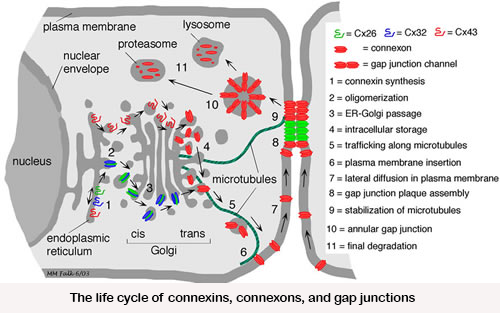 focal adhesions, hemi-desmosomes, chemical, electrical and immunological synapses, and gap junctions. We are interested in understanding how such complex signaling structures are biosynthesized, how they are structured, and how their function is regulated.
focal adhesions, hemi-desmosomes, chemical, electrical and immunological synapses, and gap junctions. We are interested in understanding how such complex signaling structures are biosynthesized, how they are structured, and how their function is regulated.
Gap junctions are the only known cellular structures that allow a direct transfer of signaling molecules from cell-to-cell by forming hydrophilic channels that bridge the opposing plasma membranes of neighboring cells. The crucial role of gap junction mediated intercellular communication (GJIC) for coordination of development, tissue function, and cell-homeostasis is now well documented; and mutations in gap junction channel protein encoding genes can result in a number of diseases, that include deafness, cataracts, severe dermatological disorders, neuropathies, cranio-facial abnormalities, and cancer. In addition, more recent findings indicate that GJIC also plays a significant role in transient cell-cell contacts, and that gap junction hemi-channels (connexons) by themselves can function in intra/extra-cellular signaling.
Biosynthesis of these channels is a complex, highly regulated process. Over the past decades we have studied where and how the gap junction channel proteins (connexins) are biosynthesized, elucidated gap junction channel subunit compatibility, characterized signals that regulate subunit interaction, determined cellular components involved in these processes, characterized how newly synthesized gap junction channels are trafficked to the plasma membrane, and have investigated how gap junctions are assembled. 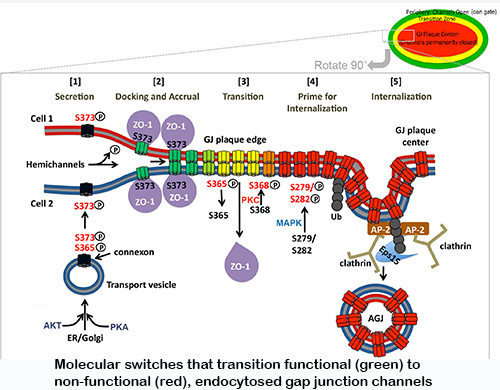 More recently, we also investigated on a molecular level how gap junctions are endocytosed and degraded. We characterized a set of post-translational modifications that occur in sequence on the connexin43 C-terminal domain that transition functional channels into non-functional channels that can interact with the clathrin-mediated endocytic machinery and become endocytosed.
More recently, we also investigated on a molecular level how gap junctions are endocytosed and degraded. We characterized a set of post-translational modifications that occur in sequence on the connexin43 C-terminal domain that transition functional channels into non-functional channels that can interact with the clathrin-mediated endocytic machinery and become endocytosed.
Modifications include the binding and release of a scaffolding protein (ZO-1), PKC and MAPK-mediated phosphorylation and de-phosphorylation events on several serine residues, and K63-poly-ubiquitination. In current projects we investigate, using molecular modeling and simulation how structurally these post-translational modifications may regulate gap junction turnover (in collaboration with the Im lab), whether modified and mutated channels have different physiological properties (in collaboration with the Haas lab), and whether impaired gap junction turnover can cause human disease (in collaboration with the Iovine lab). Interestingly, connexins and gap junctions have a very short half-live of only a few hours and are turned over constantly. Using the zebrafish model system, we could demonstrate that interfering with normal gap junction turnover causes severe developmental and functional defects, e.g., of the heart and 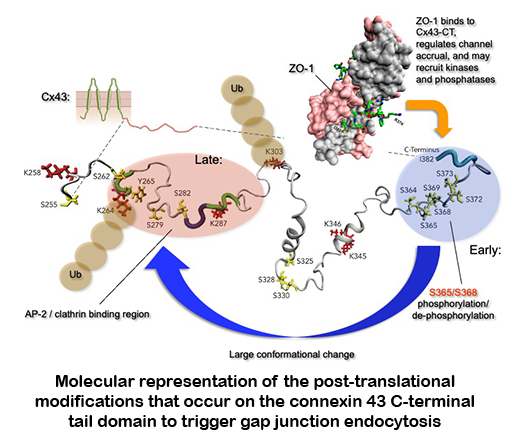 vasculature , and thus mutations naturally occurring in gap junction proteins that interfere with turnover need to be considered as potentially causing human disease. Another area of interest explores the possibility that pathogens, e.g., viruses could use internalizing gap junctions as a vehicle to move directly from cell to cell for efficient host infection.
vasculature , and thus mutations naturally occurring in gap junction proteins that interfere with turnover need to be considered as potentially causing human disease. Another area of interest explores the possibility that pathogens, e.g., viruses could use internalizing gap junctions as a vehicle to move directly from cell to cell for efficient host infection.
Our research plan is based on an integrated approach that combines novel high-resolution fluorescence and electron microscopic (in collaboration with Mark Terasaki, UConn) imaging techniques with molecular biology, biochemistry, immunological, and functional assays. Proteins are analyzed in cell-free expression systems, in cells in culture, in tissues, and in whole organisms.
Biological characterization of bioengineered tailored amorphous multi-porous (TAMP) scaffolds
My laboratory is also testing the biocompatibility and the bioactivity of dual-porous glass bone-replacement scaffolds that are produced here at Lehigh University at the “International Materials Institute for New Functionality in Glass (IMI-NFG)” headed by Prof. Himanshu Jain. Bioglass, a material that is bioactive (meaning it bonds to biological tissues), not only biocompatible (such as titanium, ceramics, and polymers that are used widely as implants) has been invented in the sixties by Larry Hentsch. TAMP scaffolds are vastly further developed bioactive glasses that have been optimized for tissue compatibility, specifically by the introduction of interconnected macro and nano porosity. Three aspects of TAMP scaffolds are particularly exciting to me: (1) These ‘spongy” materials allow the colonization with cells throughout the scaffolds while, at the same time allow the efficient dissolution of metabolic waste products; (2) TAMP materials dissolve over time in the body and thus represent true scaffolds; and (3) dissolution products have been shown to aid in the differentiation of precursor cells into mature, calcified matric secreting bone cells (osteoblasts). We have explored the use of these materials in bone and skin regeneration, dental applications, as well as in improving in vitro fertilization regiments. So far, our collaboration resulted in 11 publications and two patents.

Over 70 publications in peer reviewed international jourals and books (4,484 citations, h-index 35 as of September 2021). For a complete publication list see my profile in Google Scholar.
![]() Levin, M., Spiro, R., Jain, H. and Falk, M.M. 2022. Effects of Titanium Implant Surface topology on Bone Cell Attachment and Proliferation in vitro Medical Devices: Evidence and Research 2022:15 103-119.
Levin, M., Spiro, R., Jain, H. and Falk, M.M. 2022. Effects of Titanium Implant Surface topology on Bone Cell Attachment and Proliferation in vitro Medical Devices: Evidence and Research 2022:15 103-119.
![]() Hyland, C., Mfarej, M., Hiotis, G., Lancaster, S., Novak, N., Iovine, M.K., and Falk, M.M. 2021. Impaired Cx43 gap junction endocytosis causes morphological and functional defects in zebrafish. Mol Biol Cell Aug 11:mbcE20120797. Online ahead of print. PMID: 34379446.
Hyland, C., Mfarej, M., Hiotis, G., Lancaster, S., Novak, N., Iovine, M.K., and Falk, M.M. 2021. Impaired Cx43 gap junction endocytosis causes morphological and functional defects in zebrafish. Mol Biol Cell Aug 11:mbcE20120797. Online ahead of print. PMID: 34379446.
![]() Thamma, U., Kowal, T.J., Falk, M.M., and Jain, H. 2021. Nanostructure of bioactive glass affects bone cell attachment via protein restructuring upon adsorption. Sci Rep. 2021 Mar 11;11(1):5763. doi: 10.1038/s41598-021-85050-7. PMID: 33707489.
Thamma, U., Kowal, T.J., Falk, M.M., and Jain, H. 2021. Nanostructure of bioactive glass affects bone cell attachment via protein restructuring upon adsorption. Sci Rep. 2021 Mar 11;11(1):5763. doi: 10.1038/s41598-021-85050-7. PMID: 33707489.
![]() Liu, J., Chen, C.A., Zhu, X., Morrow, B.R., Thamma, U., Kowal, T.J., Moawad, H.M., Falk, M.M., Jain, H., and Huang, G.T. 2021. Potential of tailored amorphous multiporous calcium silicate glass for pulp capping regenerative endodontics – A preliminary assessment. J. Dent. Jun;109:103655.doi:10.1016 /j.jdent.2021.103655. PMID: 33798640.
Liu, J., Chen, C.A., Zhu, X., Morrow, B.R., Thamma, U., Kowal, T.J., Moawad, H.M., Falk, M.M., Jain, H., and Huang, G.T. 2021. Potential of tailored amorphous multiporous calcium silicate glass for pulp capping regenerative endodontics – A preliminary assessment. J. Dent. Jun;109:103655.doi:10.1016 /j.jdent.2021.103655. PMID: 33798640.
![]() Falk, M.M. 2020. Do CAR and CAR family members aid in gap junction formation? Bioessays. Dec;42(12):e2000276. doi: 10.1002/bies.202000276. PMID: 33145803.
Falk, M.M. 2020. Do CAR and CAR family members aid in gap junction formation? Bioessays. Dec;42(12):e2000276. doi: 10.1002/bies.202000276. PMID: 33145803.
![]() Bhattacharya, S., Hyland, C., Falk, M.M., and Iovine, M.K. 2020. Connexin 43 gap junctional intercellular communication inhibits evx1 expression and joint formation in regenerating fins. Development Jul 3;147(13):dev190512. doi: 10.1242/dev.190512.
Bhattacharya, S., Hyland, C., Falk, M.M., and Iovine, M.K. 2020. Connexin 43 gap junctional intercellular communication inhibits evx1 expression and joint formation in regenerating fins. Development Jul 3;147(13):dev190512. doi: 10.1242/dev.190512.
![]() Thamma, U., Kowal, T.J., Falk, M.M., and Jain, H. 2019. Influence of nanoporosity on the nature of hydroxyapatite formed on bioactive calcium silicate model glass. J. Biomed. Mater. Res. B Appl. Biomater. May;107(4):886-899.
Thamma, U., Kowal, T.J., Falk, M.M., and Jain, H. 2019. Influence of nanoporosity on the nature of hydroxyapatite formed on bioactive calcium silicate model glass. J. Biomed. Mater. Res. B Appl. Biomater. May;107(4):886-899.
![]() Kells-Andrews, R.M., Margraf, R.A., Fisher, C.G., and Falk, M.M. 2018. Connexin-43 K63-polyubiquitylation on lysines 264 and 303 regulates gap junction internalization. J. Cell Sci. 131(15):1-15.
Kells-Andrews, R.M., Margraf, R.A., Fisher, C.G., and Falk, M.M. 2018. Connexin-43 K63-polyubiquitylation on lysines 264 and 303 regulates gap junction internalization. J. Cell Sci. 131(15):1-15.
![]() Kowal, T.J., Hahn, N.C., Eider, S., Marzillier, J.Y., Fodera, D.M., Thamma, U., Jain, H., and Falk, M.M. 2018. New bioactive glass scaffolds with exceptional qualities for bone tissue regeneration: Response of osteoblasts and osteoclasts. Biomedical Materials, Jan 24; 13(2):025005. doi: 10.1088/1748-605X/aa9385. PMID: 29033393.
Kowal, T.J., Hahn, N.C., Eider, S., Marzillier, J.Y., Fodera, D.M., Thamma, U., Jain, H., and Falk, M.M. 2018. New bioactive glass scaffolds with exceptional qualities for bone tissue regeneration: Response of osteoblasts and osteoclasts. Biomedical Materials, Jan 24; 13(2):025005. doi: 10.1088/1748-605X/aa9385. PMID: 29033393.
![]() Thévenin, A.F., Margraf, R. A. Fisher, C. G., Kells-Andrews, R. M. and Falk, M.M. 2017. Phosphorylation regulates connexin43/ZO-1 binding and release, an important step in gap junction turnover. Molecular Biology of the Cell 28:3595-3608 PMID: 29021339.
Thévenin, A.F., Margraf, R. A. Fisher, C. G., Kells-Andrews, R. M. and Falk, M.M. 2017. Phosphorylation regulates connexin43/ZO-1 binding and release, an important step in gap junction turnover. Molecular Biology of the Cell 28:3595-3608 PMID: 29021339.
![]() Kowal, T.J., Golovchak, R., Chokshi, T., Harmes, J., Thamma, U., Jain, H., and Falk, M.M. 2017. Role of Phase Separation on the biological performance of 45S5 Bioglass®. J. Mater. Science: Mater. in Medicine 28:161.
Kowal, T.J., Golovchak, R., Chokshi, T., Harmes, J., Thamma, U., Jain, H., and Falk, M.M. 2017. Role of Phase Separation on the biological performance of 45S5 Bioglass®. J. Mater. Science: Mater. in Medicine 28:161.
![]() Falk, M.M., Kowal, T.J., Almaida, R.M., Saad, M., Marei, M.K., Thamma, U., and Jain, H. 2016. Chapter 5: Sol-gel glass and nano-macro porous bioscaffolds, p. 105-135. In Bioactive Glasses: Fundamentals, Technology and Applications. R. Boccaccini, D. Brauer, L. Hupa (Eds). Publisher: Royal Society of Chemistry.
Falk, M.M., Kowal, T.J., Almaida, R.M., Saad, M., Marei, M.K., Thamma, U., and Jain, H. 2016. Chapter 5: Sol-gel glass and nano-macro porous bioscaffolds, p. 105-135. In Bioactive Glasses: Fundamentals, Technology and Applications. R. Boccaccini, D. Brauer, L. Hupa (Eds). Publisher: Royal Society of Chemistry.
![]() Falk, M.M., Fisher, C.G., Kells-Andrews, R.M., and Kowal, T.J. 2016. Chapter 2: Imaging gap junctions in living cells, p. 21-62. In Gap Junction Channels and Hemichannels, Methods in Signal Transduction. D. Bai and J. C. Saez (Eds.), CRC Press, Taylor & Francis Group.
Falk, M.M., Fisher, C.G., Kells-Andrews, R.M., and Kowal, T.J. 2016. Chapter 2: Imaging gap junctions in living cells, p. 21-62. In Gap Junction Channels and Hemichannels, Methods in Signal Transduction. D. Bai and J. C. Saez (Eds.), CRC Press, Taylor & Francis Group.
![]() Falk, M.M., Bell, C.L., Kells Andrews, R.M., and Murray, S.A. 2016. Formation, Trafficking and Processing of Annular Gap Junctions. BMC Cell Biology, 17 (Suppl. 1):22.
Falk, M.M., Bell, C.L., Kells Andrews, R.M., and Murray, S.A. 2016. Formation, Trafficking and Processing of Annular Gap Junctions. BMC Cell Biology, 17 (Suppl. 1):22.
![]() Kowal, T.J. and Falk, M.M. 2015. Primary cilia found on Hela and other cancer cells. Cell Biology International 39:1341-1347.
Kowal, T.J. and Falk, M.M. 2015. Primary cilia found on Hela and other cancer cells. Cell Biology International 39:1341-1347.
![]() Nimlamool, W., Kells Andrews, R.M. and Falk, M.M. 2015. Connexin43 phosphorylation by PKC and MAPK signals VEGF-mediated gap junction internalization. Molecular Biology of the Cell 26:276-55-2768.
Nimlamool, W., Kells Andrews, R.M. and Falk, M.M. 2015. Connexin43 phosphorylation by PKC and MAPK signals VEGF-mediated gap junction internalization. Molecular Biology of the Cell 26:276-55-2768.
![]() Falk, M.M. 2015. Chapter 19: Autophagy degrades endocytosed gap junctions, p. 273-286. In Autophagy: Cancer, Other Pathologies, Inflammation, Infection, and Aging. Vol. 6, M. A. Hayat (Ed.), Academic Press, Elsevier.
Falk, M.M. 2015. Chapter 19: Autophagy degrades endocytosed gap junctions, p. 273-286. In Autophagy: Cancer, Other Pathologies, Inflammation, Infection, and Aging. Vol. 6, M. A. Hayat (Ed.), Academic Press, Elsevier.
![]() Falk, M.M., Kells, R.M., Berthoud, V.M. 2014. Degradation of connexins and gap junctions. FEBS Letters 588:1221-1229.
Falk, M.M., Kells, R.M., Berthoud, V.M. 2014. Degradation of connexins and gap junctions. FEBS Letters 588:1221-1229.
![]() Fong, J.T., Nimlamool, W., Falk, M.M. 2014. EGF induces efficient Cx43 gap junction endocytosis in mouse embryonic stem cell colonies via phosphorylation of Ser262, Ser279/282, and Ser368. FEBS Letters 588:836-844.
Fong, J.T., Nimlamool, W., Falk, M.M. 2014. EGF induces efficient Cx43 gap junction endocytosis in mouse embryonic stem cell colonies via phosphorylation of Ser262, Ser279/282, and Ser368. FEBS Letters 588:836-844.
![]() Fong, J.T., Kells, R.M., Falk, M.M. 2013. Two tyrosine-based sorting signals in the Cx43 C-terminus cooperate to mediate gap junction endocytosis. Molecular Biology of the Cell 24:2834-2848.
Fong, J.T., Kells, R.M., Falk, M.M. 2013. Two tyrosine-based sorting signals in the Cx43 C-terminus cooperate to mediate gap junction endocytosis. Molecular Biology of the Cell 24:2834-2848.
![]() Wang, S., Kowal, T.J., Marei, M.K, Falk, M.M., and Jain, H. 2013. Nanoporosity significantly enhances the biological performance of engineered glass scaffolds. Tissue Engineering Part A 19:1632-1640.
Wang, S., Kowal, T.J., Marei, M.K, Falk, M.M., and Jain, H. 2013. Nanoporosity significantly enhances the biological performance of engineered glass scaffolds. Tissue Engineering Part A 19:1632-1640.
![]() Thévenin, A.F. Kowal, T.J., Fong, J.T., Kells, R.M., Fisher, C.G., and Falk, M.M. 2013. Proteins and mechanisms regulating gap junction assembly, internalization and degradation. Physiology 28:93-116.
Thévenin, A.F. Kowal, T.J., Fong, J.T., Kells, R.M., Fisher, C.G., and Falk, M.M. 2013. Proteins and mechanisms regulating gap junction assembly, internalization and degradation. Physiology 28:93-116.
![]() Baker, S.M, and Falk, M.M. 2012. Natural inflammatory mediators thrombin and endothelin modulate gap junction intercellular communication and cell-cell adhesion. eBook. Nova Science Publishers, Inc., Hauppauge, NY, USA (ISBN: 978-1-62417-375-2).
Baker, S.M, and Falk, M.M. 2012. Natural inflammatory mediators thrombin and endothelin modulate gap junction intercellular communication and cell-cell adhesion. eBook. Nova Science Publishers, Inc., Hauppauge, NY, USA (ISBN: 978-1-62417-375-2).
![]() Falk, M.M., Fong, J.T., Kells, R.M., O'Laughlin, M.C., Kowal, T.J., Thévenin, A.F. 2012. Degradation of endocytosed gap junctions by autophagosomal and endo-/lysosomal pathways: a perspective. J. Membr. Biol. 245:465-76.
Falk, M.M., Fong, J.T., Kells, R.M., O'Laughlin, M.C., Kowal, T.J., Thévenin, A.F. 2012. Degradation of endocytosed gap junctions by autophagosomal and endo-/lysosomal pathways: a perspective. J. Membr. Biol. 245:465-76.
![]() Fong, J.T., Kells, R.M., Gumpert, A.M., Marzillier, J.Y., Davidson, M.W. and Falk, M.M. 2012. Internalized gap junctions are degraded by autophagy. Autophagy 8:794-811. (Evaluated by ‘Faculty of 1000’)
Fong, J.T., Kells, R.M., Gumpert, A.M., Marzillier, J.Y., Davidson, M.W. and Falk, M.M. 2012. Internalized gap junctions are degraded by autophagy. Autophagy 8:794-811. (Evaluated by ‘Faculty of 1000’)
Baker, S.M., and Falk, M.M. 2012. Chapter 18: Thrombin-mediated regulation of gap junction intercellular communication and cell-to-cell adhesion, pp. 247-254. In Thrombin: Function and Pathophysiology, Th. Stief (Ed.), Nova Science Publishers, Inc., Hauppauge, NY, USA (ISBN: 978-1-61942-087-8).
![]() Jain, R.H., Marzillier, J.Y., Kowal, T.J., Wang, S., Jain, H., and Falk, M.M. 2011. Expression of mineralized tissue-associated proteins is highly upregulated in MC3T3-E1 osteoblasts grown on a borosilicate glass substrate. Advances in Bioceramics and Porous Ceramics IV: Ceramic Engineering & Science Proceedings, R. Narayan & P. Colombo (Eds.) vol. 32 (6):111-122.
Jain, R.H., Marzillier, J.Y., Kowal, T.J., Wang, S., Jain, H., and Falk, M.M. 2011. Expression of mineralized tissue-associated proteins is highly upregulated in MC3T3-E1 osteoblasts grown on a borosilicate glass substrate. Advances in Bioceramics and Porous Ceramics IV: Ceramic Engineering & Science Proceedings, R. Narayan & P. Colombo (Eds.) vol. 32 (6):111-122.
![]() Wang, S., Falk, M.M., Rashad, A., Saad, M.M., Marques, A.C., Almeida, R.M., Marei, M.K., and Jain, H. 2011. Evaluation of 3D nano-macro porous bioactive glass scaffold for hard tissue engineering. J. Mater. Sci: Mater. Med. 22:1195-1203.
Wang, S., Falk, M.M., Rashad, A., Saad, M.M., Marques, A.C., Almeida, R.M., Marei, M.K., and Jain, H. 2011. Evaluation of 3D nano-macro porous bioactive glass scaffold for hard tissue engineering. J. Mater. Sci: Mater. Med. 22:1195-1203.
![]() Govindarajan, R., Chakraborty, S., Johnson, K.E., Falk, M.M., Wheelock, M.J., Johnson, K.R., and Mehta, P.P. 2010. Assembly of connexin43 into gap junctions is regulated differentially by E-cadherin and N-cadherin in rat liver epithelial cells. Mol. Biol. Cell 21:4089-4107.
Govindarajan, R., Chakraborty, S., Johnson, K.E., Falk, M.M., Wheelock, M.J., Johnson, K.R., and Mehta, P.P. 2010. Assembly of connexin43 into gap junctions is regulated differentially by E-cadherin and N-cadherin in rat liver epithelial cells. Mol. Biol. Cell 21:4089-4107.
![]() Vueva, Y., Gama, A., Almeida, R., Wang S., Jain, H., and Falk, M.M. 2010. Monolithic glass scaffolds with dual porosity prepared by polymer-induced phase separation and sol-gel. J. Americ. Ceramic Soc. 93(7):1945-1949.
Vueva, Y., Gama, A., Almeida, R., Wang S., Jain, H., and Falk, M.M. 2010. Monolithic glass scaffolds with dual porosity prepared by polymer-induced phase separation and sol-gel. J. Americ. Ceramic Soc. 93(7):1945-1949.
![]() Falk, M.M. 2010. Adherens junctions remain dynamic. BMC Biology 8:34-37.
Falk, M.M. 2010. Adherens junctions remain dynamic. BMC Biology 8:34-37.
![]() Baker, S.M., R.W. Buckheit III, and Falk, M.M. 2010. Green-to-red photoconvertible fluorescent proteins: tracking cell and protein dynamics on standard wide-field mercury arc-based microscopes. BMC Cell Biology11:15-24. (Evaluated by ‘Faculty of 1000’) (highly accessed)
Baker, S.M., R.W. Buckheit III, and Falk, M.M. 2010. Green-to-red photoconvertible fluorescent proteins: tracking cell and protein dynamics on standard wide-field mercury arc-based microscopes. BMC Cell Biology11:15-24. (Evaluated by ‘Faculty of 1000’) (highly accessed)
![]() Chakraborty, S., S. Mitra, M.M. Falk, S. Caplan, M.J. Wheelock, K.R. Johnson; and P.M. Mehta. 2010. E-Cadherin Differentially Regulates the Assembly of Connexin43 and Connexin32 into Gap Junctions in Human Squamous Carcinoma Cells. J. Biol. Chem.285:10761-10776.
Chakraborty, S., S. Mitra, M.M. Falk, S. Caplan, M.J. Wheelock, K.R. Johnson; and P.M. Mehta. 2010. E-Cadherin Differentially Regulates the Assembly of Connexin43 and Connexin32 into Gap Junctions in Human Squamous Carcinoma Cells. J. Biol. Chem.285:10761-10776.
![]() Jain, R.H., Wang, S., Moawad, H., Falk, M.M., Jain, H. 2010. Glass Bone Implants: The Effect of Surface Topology on Attachment and Proliferation of Osteoblast Cells on 45S Bioactive Glass. In Engineering Biomaterials for Regenerative Medicine; S. Bhatia, S. Bryant, J.A. Burdick, J.M. Karp, K. Walline (Eds.). Mater. Res. Soc. Symp. Proc. Vol. 1235, Warrendale, PA, 1235-RR03-47 (6 pages).
Jain, R.H., Wang, S., Moawad, H., Falk, M.M., Jain, H. 2010. Glass Bone Implants: The Effect of Surface Topology on Attachment and Proliferation of Osteoblast Cells on 45S Bioactive Glass. In Engineering Biomaterials for Regenerative Medicine; S. Bhatia, S. Bryant, J.A. Burdick, J.M. Karp, K. Walline (Eds.). Mater. Res. Soc. Symp. Proc. Vol. 1235, Warrendale, PA, 1235-RR03-47 (6 pages).
![]() Marques, A., Almaida, R., Thima, A., Falk, M.M., Jain, H. 2009. Sol-gel derived glass scaffold with high pore interconnectivity and enhanced bioactivity. J. Materials Res., 24:3495-3502.
Marques, A., Almaida, R., Thima, A., Falk, M.M., Jain, H. 2009. Sol-gel derived glass scaffold with high pore interconnectivity and enhanced bioactivity. J. Materials Res., 24:3495-3502.
![]() Moawad, H.M., Wang, S., Jain, H., Falk, M.M. 2009. Effect of zinc on bioactivity of nano-macroporous soda-lime phosphofluorosilicate glass-ceramics. Ceramic Engineering & Science Proceedings, Vol. 30, Issue 6, pp. 179-190.
Moawad, H.M., Wang, S., Jain, H., Falk, M.M. 2009. Effect of zinc on bioactivity of nano-macroporous soda-lime phosphofluorosilicate glass-ceramics. Ceramic Engineering & Science Proceedings, Vol. 30, Issue 6, pp. 179-190.
![]() Falk, M.M. S. Baker, A.M. Gumpert, D. Segretain, and R.W. Buckheit III. 2009. Gap junction turnover is achieved by the internalization of small endocytic double-membrane vesicles. Mol. Biol. Cell 20:3342-3352. (Evaluated by ‘Faculty of 1000’)
Falk, M.M. S. Baker, A.M. Gumpert, D. Segretain, and R.W. Buckheit III. 2009. Gap junction turnover is achieved by the internalization of small endocytic double-membrane vesicles. Mol. Biol. Cell 20:3342-3352. (Evaluated by ‘Faculty of 1000’)
![]() Gilleron, J., C. Fiorini, D. Carette, C. Avondet, M.M. Falk, D. Segretain, and G. Pointis. 2008. Molecular reorganization of Cx43, ZO-1, and c-Src interactions during HCH-induced internalization of gap junctions. J. Cell Sci. 121:4069-4078.
Gilleron, J., C. Fiorini, D. Carette, C. Avondet, M.M. Falk, D. Segretain, and G. Pointis. 2008. Molecular reorganization of Cx43, ZO-1, and c-Src interactions during HCH-induced internalization of gap junctions. J. Cell Sci. 121:4069-4078.
![]() Baker, S.M., N. Kim, D. Segretain, and M.M. Falk. 2008. Acute internalization of gap junctions in vascular endothelial cells in response to inflammatory mediator-induced G-protein coupled receptor activation. FEBS Lett. 582:4039-4046 (Including Journal Issue Cover).
Baker, S.M., N. Kim, D. Segretain, and M.M. Falk. 2008. Acute internalization of gap junctions in vascular endothelial cells in response to inflammatory mediator-induced G-protein coupled receptor activation. FEBS Lett. 582:4039-4046 (Including Journal Issue Cover).
![]() Gumpert, A.M., J.S. Varco, S.M. Baker, M. Piehl, and M.M. Falk. 2008. Double-membrane gap junction internalization requires the clathrin-mediated endocytic machinery. FEBS Lett. 582, 2887-2892.
Gumpert, A.M., J.S. Varco, S.M. Baker, M. Piehl, and M.M. Falk. 2008. Double-membrane gap junction internalization requires the clathrin-mediated endocytic machinery. FEBS Lett. 582, 2887-2892.
![]() Iovine, M.K., A.M. Gumpert, M.M. Falk, and T.C. Mendelsohn. 2008. Cx23, a connexin with only four extracellular-loop cysteines, forms functional gap junction channels and hemichannels. FEBS Lett. 582:165-170.
Iovine, M.K., A.M. Gumpert, M.M. Falk, and T.C. Mendelsohn. 2008. Cx23, a connexin with only four extracellular-loop cysteines, forms functional gap junction channels and hemichannels. FEBS Lett. 582:165-170.
Gilleron, J., D., Segretain, and M.M. Falk. 2007. Gap junction trafficking and regulation. Reactome: a curated knowledgebase of biological pathways. Cold Spring Harbor Laboratory, European Bioinformatics Institute, and GO Consortium.http://www.reactome.org
![]() Piehl, M., C. Lehmann, A. Gumpert, J.-P. Denizot, D. Segretain, and M.M. Falk. 2007. Internalization of large double-membrane intercellular vesicles by a clathrin-dependent endocytic process. Mol. Biol. Cell 18:337-347. (Evaluated by ‘Faculty of 1000’)
Piehl, M., C. Lehmann, A. Gumpert, J.-P. Denizot, D. Segretain, and M.M. Falk. 2007. Internalization of large double-membrane intercellular vesicles by a clathrin-dependent endocytic process. Mol. Biol. Cell 18:337-347. (Evaluated by ‘Faculty of 1000’)
![]() Eastman, S. D., T. H. Chen, M. M. Falk, T. C. Mendelson, and M. K. Iovine. 2006. Phylogenetic analysis of three complete gap junction gene families reveals lineage-specific duplications and highly supported gene classes. Genomics. 2006 Feb; 87(2):265-74.
Eastman, S. D., T. H. Chen, M. M. Falk, T. C. Mendelson, and M. K. Iovine. 2006. Phylogenetic analysis of three complete gap junction gene families reveals lineage-specific duplications and highly supported gene classes. Genomics. 2006 Feb; 87(2):265-74.
![]() Falk, M.M. 2005. Session 2: Connexin Binding Proteins. In: Laird, D. W. Overview of the 2005 International Gap Junction Conference. Published in Cell Communication and Adhesion, Vol. 6, Number 5, Dec. 2005.
Falk, M.M. 2005. Session 2: Connexin Binding Proteins. In: Laird, D. W. Overview of the 2005 International Gap Junction Conference. Published in Cell Communication and Adhesion, Vol. 6, Number 5, Dec. 2005.
![]() Segretain, D, and M.M. Falk, 2004. Regulation of connexin biosynthesis, assembly, gap junction formation, and removal. Biochim. Biophys. Acta 1662, 3-21.
Segretain, D, and M.M. Falk, 2004. Regulation of connexin biosynthesis, assembly, gap junction formation, and removal. Biochim. Biophys. Acta 1662, 3-21.
![]() Lagrée, V., K. Brunschwig, P. Lopez, N. B. Gilula, G. Richard, and M.M. Falk. 2003. Specific amino acid residues in the N-terminus and TM3 implicated in channel function and oligomerization compatibility of connexin43. J. Cell Science 116, 3189-3201.
Lagrée, V., K. Brunschwig, P. Lopez, N. B. Gilula, G. Richard, and M.M. Falk. 2003. Specific amino acid residues in the N-terminus and TM3 implicated in channel function and oligomerization compatibility of connexin43. J. Cell Science 116, 3189-3201.
![]() Falk, M. M. 2002. Genetic tags for labelling live cells: gap junctions and beyond. Trends Cell Biol. 12:399-404.
Falk, M. M. 2002. Genetic tags for labelling live cells: gap junctions and beyond. Trends Cell Biol. 12:399-404.
![]() Lauf, U., B.N.G. Giepmans, P. Lopez, S. Braconnot, S.-C. Chen, and M.M. Falk. 2002. Dynamic trafficking and delivery of connexons to the plasma membrane and accretion to gap junctions in living cells. Proc. Natl. Acad Sci. USA 99:10446-10451.
Lauf, U., B.N.G. Giepmans, P. Lopez, S. Braconnot, S.-C. Chen, and M.M. Falk. 2002. Dynamic trafficking and delivery of connexons to the plasma membrane and accretion to gap junctions in living cells. Proc. Natl. Acad Sci. USA 99:10446-10451.
(On-Line Movies at: http://www.pnas.org/cgi/content/full/162055899/DC1)
![]() Lopez, P., D. Balicki, L.K. Buehler, S.-C. Chen, and M.M. Falk. 2001. Distribution and dynamics of gap junction channels revealed in living cells. Cell Communication and Adhesion 8:237-242.
Lopez, P., D. Balicki, L.K. Buehler, S.-C. Chen, and M.M. Falk. 2001. Distribution and dynamics of gap junction channels revealed in living cells. Cell Communication and Adhesion 8:237-242.
![]() Falk, M. M. 2001. Connexins/Connexons: Cell-free expression. Methods Mol. Biol. 154:91-116.
Falk, M. M. 2001. Connexins/Connexons: Cell-free expression. Methods Mol. Biol. 154:91-116.
![]() Giepmans, B.N.G., I. Verlaan, T. Hengeveld, M.M. Falk, and W.H. Moolenaar. 2001. Gap junction protein connexin-43 interacts directly with microtubules. Current Biol. 11:1364-1368.
Giepmans, B.N.G., I. Verlaan, T. Hengeveld, M.M. Falk, and W.H. Moolenaar. 2001. Gap junction protein connexin-43 interacts directly with microtubules. Current Biol. 11:1364-1368.
Shen, P., B.N.G. Giepmans, U. Lauf, and M.M. Falk. 2001. Assembly, structure and degradation of gap junctions in living cells. In: Beat Ludin and Andrew Matus (eds.) GFP in Motion II. Trends Cell Biol. 11:183 (CD-ROM Supplement).
![]() Lauf, U., P. Lopez, and M.M. Falk. 2001. Expression of fluorescently tagged connexins: a novel approach to rescue function of oligomeric DsRed-tagged proteins. FEBS Lett. 498:11-15.
Lauf, U., P. Lopez, and M.M. Falk. 2001. Expression of fluorescently tagged connexins: a novel approach to rescue function of oligomeric DsRed-tagged proteins. FEBS Lett. 498:11-15.
![]() Falk, M.M. and U. Lauf. 2001. High-resolution fluorescence deconvolution microscopy and tagging with the autofluorescent tracers CFP, GFP, and YFP to study the structural composition of gap junctions in living cells. Microsc. Res. Tech. 52:251-262.
Falk, M.M. and U. Lauf. 2001. High-resolution fluorescence deconvolution microscopy and tagging with the autofluorescent tracers CFP, GFP, and YFP to study the structural composition of gap junctions in living cells. Microsc. Res. Tech. 52:251-262.
![]() Falk, M. M. 2000. Cell-free synthesis for analyzing the membrane integration, oligomerization, and assembly characteristics of gap junction connexins. Methods 20:165-179.
Falk, M. M. 2000. Cell-free synthesis for analyzing the membrane integration, oligomerization, and assembly characteristics of gap junction connexins. Methods 20:165-179.
![]() Falk, M.M. 2000. Connexin-specific distribution within gap junctions revealed in living cells. J. Cell Sci. 113:4109-4120.
Falk, M.M. 2000. Connexin-specific distribution within gap junctions revealed in living cells. J. Cell Sci. 113:4109-4120.
(On-Line Movies at: http://jcs.biologists.org/cgi/content/full/113/22/4109/DC1)
![]() Falk, M.M. 2000. Biosynthesis and structural composition of gap junction intercellular membrane channels. Eur. J. Cell Biol. 79:564-574.
Falk, M.M. 2000. Biosynthesis and structural composition of gap junction intercellular membrane channels. Eur. J. Cell Biol. 79:564-574.
![]() Falk, M. M. and N. B. Gilula. 1998. Connexin membrane protein biosynthesis is influenced by polypeptide positioning within the translocon and signal peptidase access. J. Biol. Chem. 273:7856-7864.
Falk, M. M. and N. B. Gilula. 1998. Connexin membrane protein biosynthesis is influenced by polypeptide positioning within the translocon and signal peptidase access. J. Biol. Chem. 273:7856-7864.
Falk, M.M., L.K. Buehler, N.M. Kumar, and N.B. Gilula. 1998. Cell-Free Expression of Functional Gap Junction Channels, p. 135-139. In R. Werner (ed.), Gap Junctions-1998. IOS Press, Amsterdam, The Netherlands.
![]() Yeager, M., V. Unger, and M.M. Falk. 1998. Synthesis, Assembly, and Structure of Gap Junction Intercellular Channels. Curr. Opin. Struc. Biol. 8:517-524.
Yeager, M., V. Unger, and M.M. Falk. 1998. Synthesis, Assembly, and Structure of Gap Junction Intercellular Channels. Curr. Opin. Struc. Biol. 8:517-524.
![]() Kahn, T.W., R.N. Beachy, and M.M. Falk. 1997. Cell-free expression of a GFP fusion protein allows quantitation in vitro and in vivo. Current Biol. 7:R207-R208.
Kahn, T.W., R.N. Beachy, and M.M. Falk. 1997. Cell-free expression of a GFP fusion protein allows quantitation in vitro and in vivo. Current Biol. 7:R207-R208.
![]() Falk, M.M., L.K. Buehler, N.M. Kumar, and N.B. Gilula. 1997. Cell-free Synthesis and Assembly of Connexins into Functional Gap Junction Membrane Channels. EMBO J. 16:2703-2716.
Falk, M.M., L.K. Buehler, N.M. Kumar, and N.B. Gilula. 1997. Cell-free Synthesis and Assembly of Connexins into Functional Gap Junction Membrane Channels. EMBO J. 16:2703-2716.
Falk, M.M., N.M. Kumar, and N.B. Gilula. 1995. Biosynthetic Membrane Integration of Connexin Proteins. Progress in Cell Research 4:319-322.
![]() Falk, M.M., N.M. Kumar, and N.B. Gilula. 1994. Membrane Insertion of Gap Junction Connexins: Polytopic Channel Forming Membrane Proteins. J. Cell Biol. 127:343-355.
Falk, M.M., N.M. Kumar, and N.B. Gilula. 1994. Membrane Insertion of Gap Junction Connexins: Polytopic Channel Forming Membrane Proteins. J. Cell Biol. 127:343-355.
![]() Falk, M.M., F. Sobrino, and E. Beck. 1992. VPg gene Amplification Correlates with Infective Particle Formation in Foot-and-Mouth Disease Virus. J. Virol. 66:2251-2260.
Falk, M.M., F. Sobrino, and E. Beck. 1992. VPg gene Amplification Correlates with Infective Particle Formation in Foot-and-Mouth Disease Virus. J. Virol. 66:2251-2260.
Falk, M.M. 1991, p. 34, Fig. 1.1.15, A. In P. Sitte, H. Ziegler, F. Ehrendorfer, and A. Bresinsky (eds.), Strasburger, Lehrbuch der Botanik, 33. Neubearbeitete Auflage-1991. Gustav Fischer Verlag, Stuttgart, Jena, New York.
![]() Zibert, A., G. Maass, K. Strebel, M.M. Falk, and E. Beck. 1990. Infectious Foot-and-Mouth Disease Virus Derived from a Cloned Full-Length cDNA. J. Virol. 64:2467-2473.
Zibert, A., G. Maass, K. Strebel, M.M. Falk, and E. Beck. 1990. Infectious Foot-and-Mouth Disease Virus Derived from a Cloned Full-Length cDNA. J. Virol. 64:2467-2473.
Falk, M.M., P.R. Grigera, I.E. Bergmann, and E. Beck. 1990. Presumed Transcription Regulating Domain of Histone H3 is Cleaved Off During Infection by Foot-and-Mouth Disease Virus. Internat. J. Microbiol. Hygiene. 272:366-367.
Falk, M.M., P.R. Grigera, I.E. Bergmann, A. Zibert, and E. Beck. 1990. Viral Protease 3C-Mediated Cleavage of Histone H3 in Foot-and-Mouth Disease Virus-Infected Cells, p. 184-187. In M. A. Brinton and F. X. Heinz (eds.), -1990. American Society for Microbiology, Washington, DC, USA.
![]() Falk, M.M., P.R. Grigera, I.E. Bergmann, A. Zibert, G. Multhaup, and E. Beck. 1990. Foot-and-Mouth Disease Virus Protease 3C Induces Specific Proteolytic Cleavage of Host Cell Histone H3. J. Virol. 64:748-756.
Falk, M.M., P.R. Grigera, I.E. Bergmann, A. Zibert, G. Multhaup, and E. Beck. 1990. Foot-and-Mouth Disease Virus Protease 3C Induces Specific Proteolytic Cleavage of Host Cell Histone H3. J. Virol. 64:748-756.
![]() Mayer, T., T. Tamura, M.M. Falk, and H. Niemann. 1988. Membrane Integration and Intracellular Transport of the Coronavirus Glycoprotein E1, a Class III Membrane Glycoprotein. J. Biol. Chem. 263:14956-14963.
Mayer, T., T. Tamura, M.M. Falk, and H. Niemann. 1988. Membrane Integration and Intracellular Transport of the Coronavirus Glycoprotein E1, a Class III Membrane Glycoprotein. J. Biol. Chem. 263:14956-14963.
Patents
“Bioactive glass preparation and use”. U.S. Patent No.: US 9,757,417 B2. Inventors: Jain, H., Falk, M.M., Holovchak, R. Patent awarded Sept. 12, 2017.
“Scaffolds for uterine cell growth”, U.S. Patent No.: US 10,907,132 B2. Inventors: Barmat, L., Falk, M.M., Jain, H., Somkuti, S. Patent awarded Feb. 2, 2021
| 12/17-12/19 | Program Director, National Science Foundation (NSF) Directorate: Biology (BIO), Division: Molecular and Cell Biology (MCB), Cluster: Cellular Dynamics and Function (CDF) |
| Since 5/16 | Professor, Department of Biological Sciences, Lehigh University, Bethlehem, Pennsylvania, USA |
| 5/09-5/16 | Associate Professor (with Tenure), Department of Biological Sciences, Lehigh University, Bethlehem, Pennsylvania, USA |
| 8/03-5/09 | Assistant Professor (Tenure Track), Department of Biological Sciences, Lehigh University, Bethlehem, Pennsylvania, USA |
| 2/98-8/03 | Assistant Professor (Non-Tenure Track), Department of Cell Biology, The Scripps Research Institute, La Jolla, California, USA |
| 7/96-2/98 | Senior Research Associate, Department of Cell Biology, The Scripps Research Institute, La Jolla, California, USA |
| 4/92-7/96 | Postdoctoral Fellow, Department of Cell Biology, The Scripps Research Institute, La Jolla, California, USA Sponsor: Dr. Norton B. Gilula Project title: Structure and function of gap junction membrane channels |
| 12/91 | Ph.D. Defense, Curriculum in Molecular Biology, Cell Biology, Virology, and Biochemistry, University of Heidelberg, Heidelberg, Germany |
| 11/87-2/92 | Ph.D. thesis, Center for Molecular Biology (ZMBH), University of Heidelberg, Heidelberg, Germany Thesis Advisor: Dr. E. Beck; chair: Dr. H. Schaller Thesis project: Structure and function of foot-and-mouth disease virus (Picornaviridae) proteins |
| 11/87 | Diploma in Biology (M.S.), University of Giessen, Giessen, Germany |
| 10/86-11/87 | Diploma thesis, Department of human virology, University of Giessen, Giessen, Germany Thesis Advisor: Dr. H. Niemann, chair: Dr. H. Bauer Thesis project: Characterization of Golgi retention signals in the E1 transmembrane glycoprotein of MHV (Coronaviridae) |
| 5/84 | Vor-Diplom in Biology (B.S.), University of Giessen, Giessen, Germany |
| 10/82-10/86 | Studied Biology at the University of Giessen, Giessen, Germany |
Click here for a recent sample syllabus and Experiment Schedule
BioS 421 (Molecular Cell Biology I)
Click here for a recent sample syllabus including topics and list of discussed papers
BioS 41 (Core I: Cell & Molecular)
Click here for a recent sample syllabus
Click the image below to see the full size document

Studying Scientific Surprises - focus article on Caitlin Hyland (Ph.D. 2021)

Falk Lab Group 2010
Falk Lab Group 2013

Falk Lab Group 2019 - a few days before COVID shut us down
Zoom lab meeting during COVID. Falk lab t-shirts for all to keep up our spirits!

The Falk Lab met with Congressman Charlie Dent

The Falk Lab met with Nobel Prize winner Martin Chalfie, Ph.D.

Raina Jain meets with President Obama

Tia Kowal earns her Ph.D.

Rachael Kells gets hooded!

Rachel Margraf, Falk Lab undergraduate research first author interview in Journal of Cell Science
Click here to read

Dr. Falk in his office at the National Science Foundation (NSF) headquarters in Alexandria, VA. (2017-2019)
Caitlin Hyland successfully defended her Ph.D. thesis (May 26, 2021)
- Lehigh Research Review 2017: Mapping Cellular Gap Junctions to Understand Disease
- Falk presents research in Cairo (2014, page 5)
- Falk works to save energy
Lehigh Bulletin, Winter 2014 (page 15)
Going Solar - Falk's microscopic art highlighted in 2012 calendar (2013, page 3)