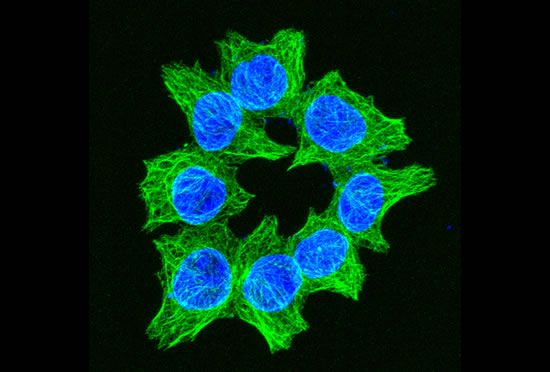
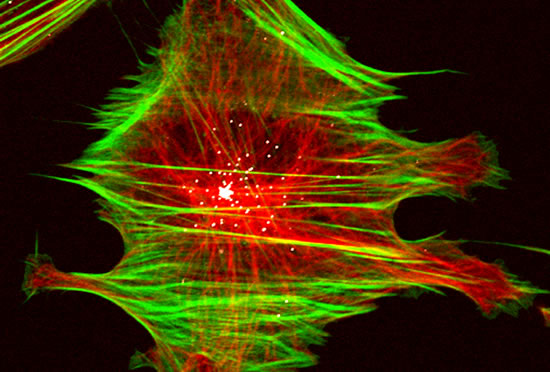
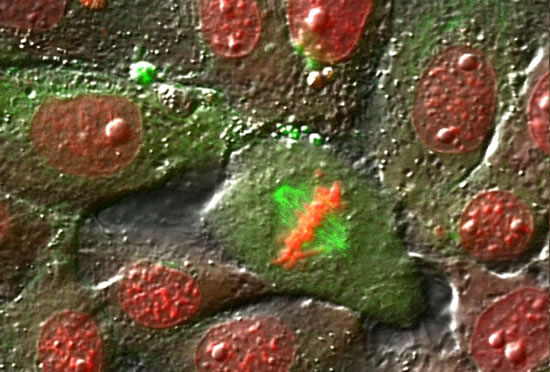
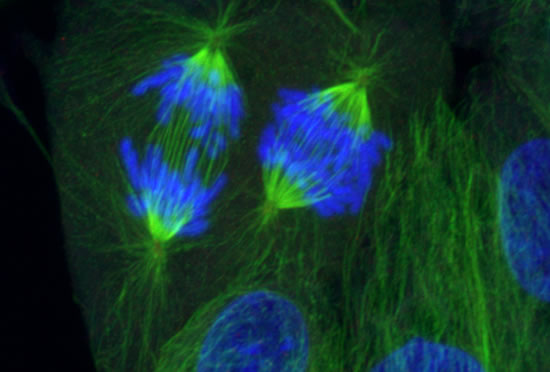
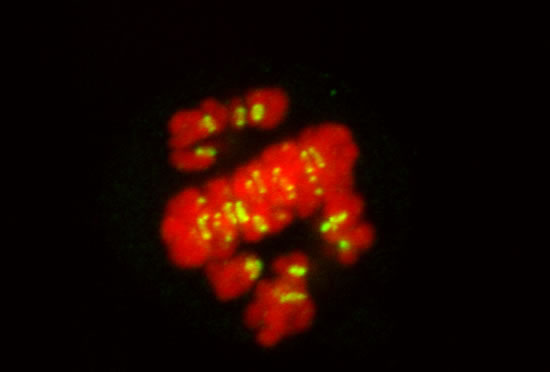
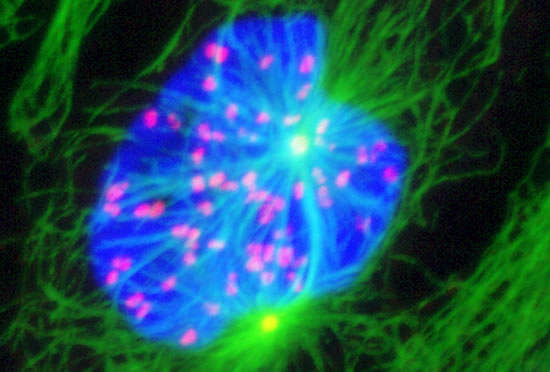
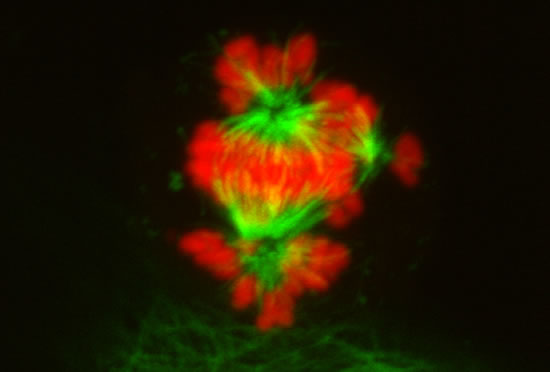
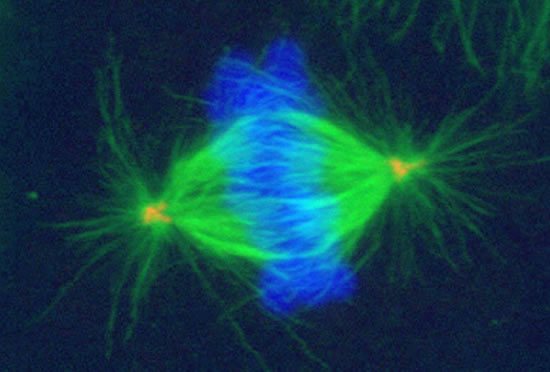
These projects are winding down after 26 years.
Click here for our latest endeavors
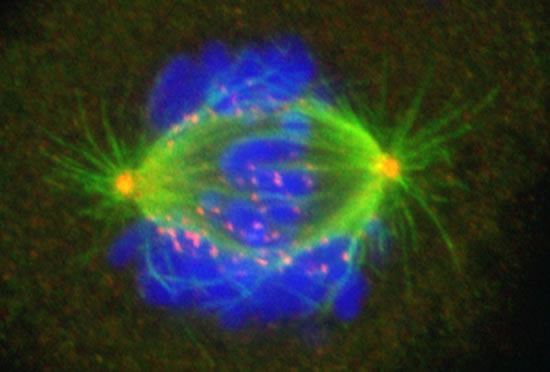
The Cassimeris lab group is interested in how animal cells proliferate - how cells increase in numbers through growth and division. Cell proliferation underlies processes such as development, where a single fertilized egg grows and divides into the trillions of cells of the adult, as well as the uncontrolled growth and division of cancer cells. Unraveling the mechanisms responsible for cell growth, division and survival will allow biologists to control cell proliferation in time or space, for example blocking cancer cell proliferation or controlling stem cell growth. Our work focuses on the cell's internal skeleton, built of polymers that assemble and disassemble every minute. These polymers (microtubules) are responsible for separating the genome into each daughter cell at each cell division and are the target of some of the most successful anti-cancer drugs.
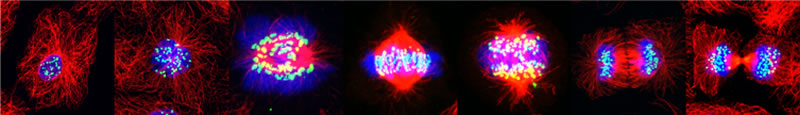
Image series by Bret Becker, Ph.D. (microtubules in red, DNA in blue, kinetochores in green/white)

J. Leung, M. Plooster, L. Cassimeris. 2016. Exploring stathmin control of cell survival through negative feedback of a JNK-dependent pathway. Matters. doi: 10.19185/matters.201602000021.
Lewis, A., M. McDonald, S. Scharbach, S. Hamaway, M. Plooster, K. Peters, K.M. Fox, L. Cassimeris, J.M. Tanski and L.A. Tyler. 2016. The chemical biology of Cu(II) complexes with imidazole or thiazole containing ligands: Synthesis, crystal structures and comparative biological activity. J. Inorg. Biochem. 157: 52 - 61.
V. C. Silva, M. Plooster, J. C. Leung and L. Cassimeris. 2015. A delay prior to mitotic entry triggers caspase 8-dependent cell death in p53-deficient Hela and HCT-116 cells. Cell Cycle. 14: 1070-1081.
R. A. Steiner, D. Foreman, X. H. Lin, B. K. Carney, K. M. Fox, L. Cassimeris, J. M. Tanski and L. A. Tyler. 2014. Synthesis, characterization, crystal structures and biological activity of set of Cu(II) benzothiazole complexes: artificial nucleases with cytotoxic activities. J. Inorganic Biochem. 137: 1-11.
Victoria C. Silva and Lynne Cassimeris. 2014. CAMSAPs add to the growing microtubule minus-end story. Dev Cell. 28: 221 - 222.
N.T. Thomas, R. Thamkachy, K.C. Sivakumar, S.K. Jayakumari, X.L. Louis, S.A. Thomas, R. Kumar, K.N. Rajasekharan, L. Cassimeris and S. Sengupta. 2014. Reversible Action of Diaminothiazoles in Cancer Cells is Implicated by the Induction of a Fast Conformational Change of Tubulin and Suppression of Microtubule Dynamics. Mol. Cancer Ther. 13: 179-189.
V. Caruso Silva and L. Cassimeris. 2013. Stathmin and microtubules regulate mitotic entry in Hela cells by controlling activation of both Aurora Kinase A and Plk1. Mol. Biol. Cell. 24: 3919-3931.
L. Cassimeris, L. Guglielmi, V. Denis, C. Larroque and P. Martineau. 2013. Specific in vivo labeling of tyrosinated a-tubulin and measurement of microtubule dynamics using a GFP tagged, cytoplasmically expressed recombinant antibody. PLoS One. 8:e59812.
S. Nakamura, I. Grigoriev, T. Nogi, T. Hamaji, L. Cassimeris and Y. Mimori-Kiyosue. 2012. Dissecting the nanoscale distributions and functions of microtubule-end-binding proteins EB1 and ch-TOG in interphase HeLa cells. PLoS One. 7: e51442.
Bruce K. Carney, V. Caruso Silva and L. Cassimeris. 2012. The microtubule cytoskeleton is required for a G2 cell cycle delay in cancer cells lacking stathmin and p53. Cytoskeleton. 69: 278-289.
Cassimeris, L., V. Caruso Silva, E. Miller, Q. Ton, C. Molnar, and J. Fong. 2012. Fueled by Microtubules: Does tubulin dimer/polymer partitioning regulate intracellular metabolism? Cytoskeleton. 69:133-143.
Blake-Hodek, K.A., L. Cassimeris and T.C. Huffaker. 2010. Regulation of microtubule dynamics by Bim1 and Bik1, the budding yeast members of the EB1 and CLIP-170 families of plus-end tracking proteins. Mol. Biol. Cell. 21: 2013 - 2023.
B.K. Carney and L. Cassimeris. 2010. Stathmin/oncoprotein 18, a microtubule regulatory protein, is required for survival of both normal and cancer cell lines lacking the tumor suppressor, p53. Cancer Biol. and Therapy. 9: 699-709.
Ringhoff, D.N. and L. Cassimeris. 2009. Gene expression profiles in mouse embryo fibroblasts lacking stathmin, a microtubule regulatory protein, reveal changes in the expression of genes contributing to cell motility. BMC Genomics. 10:343.
Ringhoff, D.N. and L. Cassimeris. 2009. Stathmin regulates centrosomal nucleation of microtubules and tubulin dimer/polymer partitioning. Mol. Biol. Cell. 20: 3451-3458.
Cassimeris, L., B. Becker and B. Carney. 2009. TOGp Regulates Microtubule Assembly and Density during Mitosis and Contributes to Chromosome Directional Instability. Cell Motil. Cytoskel. 66: 535-545.
Cassimeris, L. 2009. Microtubule Assembly: Lattice GTP to the Rescue. Curr. Biol. 19: R174 - R176.
Cassimeris, L. 2009. “Microtubule Associated Proteins in Neurons”, The New Encyclopedia of Neuroscience. (L.R. Squire, Editor). Oxford: Academic Press. 5: 865-870.
Skibbens, R.V., D.N. Ringhoff, J. Marzillier, L. Cassimeris, and L. Eastman. 2008. Positional analysis of BRCA1-dependent expression in Saccharomyces cerevisiae. Cell Cycle. 7: 3928 – 3934.
Cassimeris, L. 2009. Microtubule Associated Proteins in Neurons. The New Encyclopedia of Neuroscience. (L.R. Squire, Editor). Oxford: Academic Press. 5: 865-870.
Mehiri, M., B. Jin, D. Ringhoff, V. Janout, L. Cassimeris and S.L. Regen. 2008. Cellular entry and nuclear targeting by a highly anionic molecular umbrella. Bioconjugate Chem. 19: 1510-1513.
Bartolini, F., J.B. Moseley, J. Schmoranzer, L. Cassimeris, B.L. Goode and G.G. Gundersen. 2008. The formin mDia2 stabilizes microtubules independently of its actin nucleation activity. J. Cell Biol. 181: 523 – 536.
Cassimeris, L. 2007. Tubulin delivery: Polymerization chaperones for microtubule assembly? Dev. Cell. 13: 455 – 456.
Warren, James C. and L. Cassimeris. 2007. The Contributions of Microtubule Stability and Dynamic Instability to Adenovirus Nuclear Localization Efficiency. Cell Motil. Cytoskel. in press.
M. Murphy and L. Cassimeris. 2006. A novel cancer therapy approach targeting microtubule function. Cancer Biol. Ther. 5: 1721 – 1723.
J. Warren, A. Rutkowski and L. Cassimeris. 2006. Infection with Replication-Deficient Adenovirus Induces Changes in the Dynamic Instability of Host Cell Microtubules. Mol. Biol. Cell. 17: 3557 – 3568.
L. Cassimeris. 2006. Mitosis: Riding the protofilament curl. Current Biology. 16: R214-R216.
Becker, B.E. and L. Cassimeris. 2005. Cytoskeleton: Microtubules Born on the Run. Current Biology. 15: R551 – R554.
VanBuren, V., L. Cassimeris and D.J. Odde. 2005. A mechanochemical model of microtubule structure and self-assembly kinetics. Biophysical J. 89: 2911- 2926.
Bartolini, F., G. Tian, M. Piehl, L. Cassimeris, S.A. Lewis and N. J. Cowan. 2005. Identification of a novel tubulin-destabilizing protein related to the chaperone cofactor E. J. Cell Science. 118: 1197 – 1207.
Cassimeris. L. 2004. Cell Division: Eg’ing on Microtubule Flux. Curr. Biol. 14: R1000-R1002.
Piehl, M. U.S. Tulu, P. Wadsworth and L. Cassimeris. 2004. Centrosome maturation: Measurement of microtubule nucleation throughout the cell cycle using GFP tagged EB1. Proceedings of the National Academy of Sciences, USA. 101: 1584-1588.
Cassimeris, L. and J. Morabito. 2004. TOGp, the human homolog of XMAP215/Dis1, is required for centrosome integrity, spindle pole organization and bipolar spindle assembly. Mol. Biol. Cell. 15: 1580-1590.
Bellows, A., M. Kenna, L. Cassimeris and R.V. Skibbens. Human EFO1p exhibits acetyltransferase activity and is a unique combination of linker histone and Ctf7p/Eco1p chromatid cohesion establishment domains. Nucleic Acids Research. 31:6334 – 43.
Cassimeris, L. and R.V. Skibbens. Regulated assembly of the mitotic spindle: A perspective from two ends. Current Issues in Molecular Biology. 5: 99 - 112.
Piehl, M. and Cassimeris, L. 2003. Organization and dynamics of growing microtubule plus ends during early mitosis. Mol. Biol. Cell. 14: 916 - 925.
Segerman, B., P. Holmfeldt, J. Morabito, L. Cassimeris and M. Gullberg. 2003. Autonomous and phosphorylation-responsive microtubule-regulating activities of the N-terminus of Op18/stathmin. Journal of Cell Science.116:197-205.
V. VanBuren, D. Odde and L. Cassimeris. 2002. Estimates of Lateral and Longitudinal Bond Energies within the Microtubule Lattice. Proceedings of the National Academy of Sciences, USA. 99: 6035 - 6040.
Present Lab Members
Lynne Cassimeris, Ph.D.
Principle Investigator
email Dr. Cassimeris
Jessica Leung
Graduate Student
email Ms. Leung

Former Lab Mates
Visiting Scientist
Dr. Suparna Sengupta, Fall 2007
Home institution: Rajiv Gandhi Centre for Biotechnology, Trivandrum, India
Supported by a UICC Yamagiwa-Yoshida Memorial International Cancer Study Grant, awarded by the International Union Against Cancer
Post-doctoral research associates
Dr. Bret Becker, 2005-2007
Dr. Bruce Carney, 2012
Dr. Yagmur Micoogullari, 2016
Dr. Clare DaSilva Santos, 2016-17
Graduate Students
Ph.D.'s
Robert Vasquez, Ph.D., 1996
Bonnie Howell, Ph.D., 1998
Cynthia Spittle, Ph.D., 2000
Vincent VanBuren, Ph.D., 2001
Michelle Kratzer Piehl, Ph.D., 2003
James Warren, Ph.D., 2007
Danielle Ringhoff, Ph.D., 2009
Bruce Carney, Ph.D., 2011
Victoria Caruso Silva, Ph.D., 2014Master's
Justin Morabito, M.S., 20014
Vincent Fong, M.S., 2004
Balajirah Indrajith, M.S., 2014
Cody Molnar, M.S., 2014

Undergraduate Students
Krista Kupres, 1994-1995
Karen Rose, 1995
Eric Guelaff, 1995
Misty Saracino, 1997
Alexander Zodiatis, 1995 -1997
Jessica Lehoczky, 1997
David Stachura, 1999-2000
A. Gene Smith, 1999-2001
Jeremy McGarvey, 2003
Vincent Fong, 2003
Adam Rutkowski, 2005-2006
Kyle Keating, 2005
Alexsandra Fridshtand, 2005-2006
Areeb Zamir, 2007-2010
Morgan Lewis, 2010
Dan Ciccone, 2012-2013
Melissa Plooster, 2012-2014
Kyle Peters, 2013-2015
Arianna Caruso, 2013-2015
Jessica Pryoer, 2015
Audrey Hla, 2015
Caitlyn Wilson, 2016-2017
Chelsea Hipwell, 2017
Virtual Lab Tour
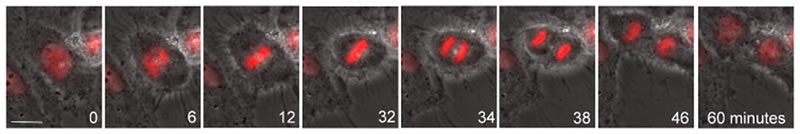
Example of Cell Division
The following video won 2nd prize at Celldance 2012, part of the annual meeting of the American Society for Cell Biology.