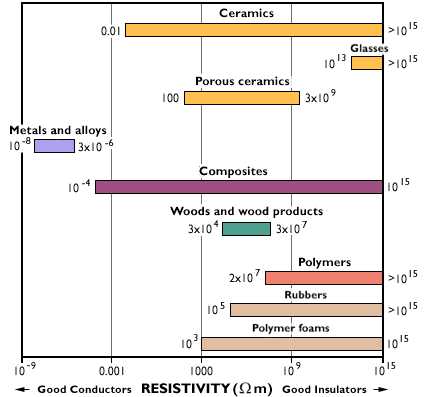
| Thermal conductivity
tells us the ease upon which thermal energy (heat for most purposes) can
move through a material. Some materials like metals allow heat to travel
through them quite quickly. Imagine that with one hand you are touching
a piece of metal and with the other, a piece of wood. Which material would
feel colder? If you said, "metal," you would be correct. But,
in fact, both materials are in fact the same temperature. This is relative
thermal conductivity. Metal has a higher heat transferability, or thermal
conductivity, than wood, letting the heat from your hand leave faster. If
you want to keep something cold the best idea is to wrap it in something
that does not have a high heat transferability, or high thermal conductivity,
this would be an insulator. Ceramics, and polymers are usually good insulators,
but you have to remember that polymers usually have a very low melting temperature.
That means if you are designing something that will get very hot the polymer
might melt, depending on its melting temperature. |
![]()
![]()